Abstract
Introduction: In our phase II study (Moskowitz AJ et al, JCO 2021), pembrolizumab plus gemcitabine, vinorelbine, and liposomal doxorubicin (P-GVD) was found to be highly effective as salvage for patients (pts) with relapsed or refractory (R/R) classic Hodgkin lymphoma (cHL) proceeding to high dose therapy and autologous hematopoietic cell transplantation (AHCT). We present analyses assessing immunologic changes associated with P-GVD treatment as well as updated clinical outcome.
Methods: Transplant eligible pts with R/R cHL following failure of 1-line of therapy were eligible. Treatment consisted of 2 to 4 cycles of P-GVD followed by AHCT. Peripheral blood samples were collected at baseline, after 2 cycles of P-GVD, before AHCT, 3 and 9 months after AHCT. Peripheral blood was analyzed for serum thymus and activation-regulated chemokine (TARC) and immune cell subsets. TARC was measured by enzyme-linked immunosorbent assay. Immune cell subsets, including T-cells and myeloid-derived suppressor cells (MDSC), were measured by multi-parametric flow cytometry (FC). Metabolic tumor volume (MTV) was evaluated at baseline. Changes in MDSC and TARC levels were assessed using Wilcoxon rank sum and Friedman tests, respectively. Correlations between MTV and either TARC or MDSC were assessed using Pearson's R.
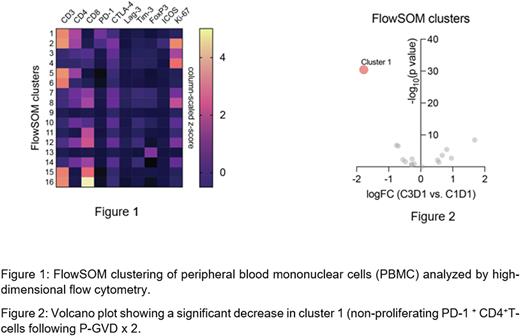
Results: Of 38 evaluable pts, the complete response (CR) rate after 2 or 4 cycles of P-GVD was 95%. Two pts declined AHCT and 36 proceeded to AHCT. After a median follow-up of 30 months (range: 2-43), only 1 pt experienced disease progression 23 months after AHCT. The estimated 30-month progression-free survival (PFS) is 96%. The median baseline MTV value was 37 cm3 (range 4- 845). 36/38 and 27/38 pts had sufficient correlative samples to perform analysis for TARC and MDSC analysis, respectively. The median baseline TARC level was 526 pg/mL (range: 12-4017). TARC levels decreased significantly after 2 cycles of P-GVD (p < 0.001) and increased post-AHCT. Circulating MDSCs were 0.05% of total mononuclear cells (range: 0.003-0.59%) and significantly increased after 2 cycles of P-GVD (p<0.001). Higher baseline TARC and circulating MDSC levels correlated with higher baseline MTV (r=0.45, 95% CI: 0.11-0.70, p<0.01 and r=0.46, 95%CI 0.03-0.76, p=0.03, respectively). High-dimensional analysis of FC data revealed several distinct immune clusters (Figure 1), of which non-proliferating PD-1+ CD4+ T-cells decreased most significantly following treatment (Figure 2). Additional markers (ICOS, CTLA-4, LAG-3, FOXP3) of immune exhaustion showed only a mild decrease over the treatment course.
Conclusions: After a median of 30-months follow-up, second-line P-GVD continues to be associated with durable remissions after AHCT. These analyses provide insights into key immune changes associated with chemoimmunotherapy in HL. Baseline TARC correlated with baseline MTV and was significantly lower after P-GVD, illustrating its role as a marker of tumor bulk. The subsequent rise in serum TARC post AHCT suggests immune reconstitution of TARC-secreting cells. Higher circulating levels of MDSCs correlated with baseline MTV, which may reflect an association between immune suppression and tumor bulk. MDSC levels unexpectedly increased after P-GVD, indicating that a rise in MDSCs after treatment may not necessarily predict for poor outcomes. Ongoing single-cell RNA sequencing analysis of peripheral blood mononuclear cells (PBMC) may provide further understanding into immune dynamics associated with P-GVD treatment.
Disclosures
Wills:Bristol Myers Squib Foundation: Research Funding. Shah:Janssen: Research Funding; Amgen: Research Funding; Beyond Spring: Research Funding. Wong:Leap Therapeutics.: Honoraria. Dogan:Incyte: Consultancy; Loxo: Consultancy; Seattle Genetics: Consultancy; Peer View: Honoraria; Takeda: Other: Research Funding; Roche: Other: Research Funding; EUSA Pharma: Consultancy; Physicians’ Education Resource: Consultancy, Honoraria. Straus:Seagen: Consultancy; Takeda Pharmaceuticals: Consultancy. Noy:Janssen: Research Funding. Kumar:BridgeBio Pharmaceuticals: Current equity holder in publicly-traded company; Abbvie: Research Funding; Adaptive Biotechnologies: Research Funding; Celgene: Research Funding; Pharmacyclics: Research Funding; Seattle Genetics: Research Funding; AstraZeneca: Honoraria, Research Funding; Kite Pharma: Honoraria; Janssen: Honoraria. Palomba:Notch Therapeutics: Current holder of stock options in a privately-held company, Honoraria; GSK: Honoraria; Da Volterra: Honoraria; Thymofox: Honoraria; Ceramedix: Honoraria; Garuda: Honoraria; Lygenesis: Honoraria; Pluto Therapeutics: Current holder of stock options in a privately-held company, Honoraria; Nektar Therapeutics: Honoraria; Frazier Healthcare Partners: Honoraria; Rheos: Honoraria; Vor Biopharma: Honoraria; Seres: Current holder of stock options in a privately-held company, Honoraria, Research Funding; MustangBio: Honoraria; Kite: Honoraria; BeiGene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Synthekine: Honoraria; Novartis: Honoraria; BMS: Consultancy. Falchi:ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy, Research Funding; Genetech: Consultancy, Research Funding; Roche: Consultancy, Research Funding; Genmab: Consultancy, Research Funding. Lahoud:MorphoSys, Inc: Membership on an entity's Board of Directors or advisory committees. Zelenetz:MBS: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Genentech/Roche: Consultancy, Honoraria, Research Funding; Juno Pharmaceuticals: Consultancy, Honoraria; Gilead/Kite Pharma: Consultancy, Honoraria; Beigene: Consultancy, Honoraria, Research Funding; Pharmacyclics/Abbvie: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; AstraZeneca: Consultancy, Honoraria; Novartis: Consultancy, Honoraria. Batlevi:Xynomic: Research Funding; Autolus: Research Funding; ADC Therapeutics: Other: Provision of Services; Juno/Celgene: Consultancy; Bristol-Myers Squibb: Other: Ownership / Equity Interests; Provision of Services; GLG Pharma: Consultancy; Dava Oncology: Other: Provision of Services; Bayer: Research Funding; Janssen: Research Funding; Novartis: Research Funding; Epizyme: Research Funding; Roche/Genentech: Research Funding; Seattle Genetics: Consultancy; Kite Pharma: Consultancy; Life Sciences: Consultancy. Horwitz:Celgene: Research Funding; SecuraBio: Honoraria; Shoreline Biosciences, Inc.: Membership on an entity's Board of Directors or advisory committees; ONO Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy; Daiichi Sankyo: Research Funding; Crispr Therapeutics: Research Funding; Verastem/SecuraBio: Research Funding; Kyowa Hakko Kirin: Research Funding; Yingli Pharma Limited and Tubulis: Honoraria; ADC Therapeutics: Research Funding; Affimed: Research Funding; Seattle Genetics,: Research Funding; C4: Research Funding; Kyowa Hakko Kirin: Consultancy; Daiichi Sankyo: Membership on an entity's Board of Directors or advisory committees; Cimieo Therapeutics: Honoraria; Affimed,: Consultancy; Millennium /Takeda: Research Funding. Matasar:Teva: Consultancy; Genentech, Inc.: Consultancy, Honoraria, Research Funding; Rocket Medical: Consultancy, Research Funding; Seattle Genetics: Consultancy, Honoraria, Research Funding; AstraZeneca: Consultancy; Juno Therapeutics: Consultancy; F. Hoffmann-La Roche Ltd.: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria; Epizyme: Consultancy, Honoraria; IGM Biosciences: Research Funding; IMV Therapeutics: Consultancy, Honoraria; TG Therapeutics: Consultancy; Karyopharm: Consultancy; ADC Therapeutics: Consultancy, Honoraria; Merck: Consultancy, Current equity holder in private company; Janssen: Honoraria, Research Funding; ImmunoVaccine Technologies: Honoraria, Research Funding; Bayer: Consultancy, Honoraria, Research Funding; Pharmacyclics: Honoraria, Research Funding; Daiichi Sankyo: Consultancy; GlaxoSmithKline: Honoraria, Research Funding. Salles:Roche/Genentech, Gilead Sciences, Janssen, Celgene, Novartis, MorphoSys AG, Epizyme, Alimera Sciences, Genmab, Debiopharm Group, Velosbio, Bristol-Myers Squibb, BeiGene, Incyte, Miltenyi Biotec, Ipsen, Kite, a Gilead Company, Loxo, Rapt: Consultancy; Roche/Genentech, Janssen, Celgene, Gilead Sciences, Novartis, AbbVie, MorphoSys AG, Amgen, Bayer, Epizyme, Regeneron, Kite, a Gilead Company: Honoraria; AbbVie, BeiGene, Bristol Myers Squibb, Celgene, Debiopharm, Epizyme, Genentech/Roche, Genmab, Incyte, Kite, a Gilead Company, Miltenyi, MorphoSys, Takeda, and VelosBio: Membership on an entity's Board of Directors or advisory committees. Moskowitz:Merck: Honoraria. Vardhana:Koch Disruptive Technologies: Consultancy; Immunai: Membership on an entity's Board of Directors or advisory committees. Moskowitz:ADC Therapeutics: Research Funding; Biegene: Research Funding; Miragen: Research Funding; Seattle Genetics: Research Funding; Merck: Research Funding; Bristol-Myers Squibb: Research Funding; Incyte: Research Funding; SecuraBio: Research Funding; Affimed: Honoraria; Imbrium Therapeutics L.P./Purdue: Honoraria; Janpix Ltd: Honoraria; Merck: Honoraria; Seattle Genetics: Honoraria; Takeda: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal